
If the parent search revealed a molecule-, cell-, tissue-, or organ-level factor linking the BBB and systemic inflammation, the factor was searched separately. In summary, a multistep approach was used. 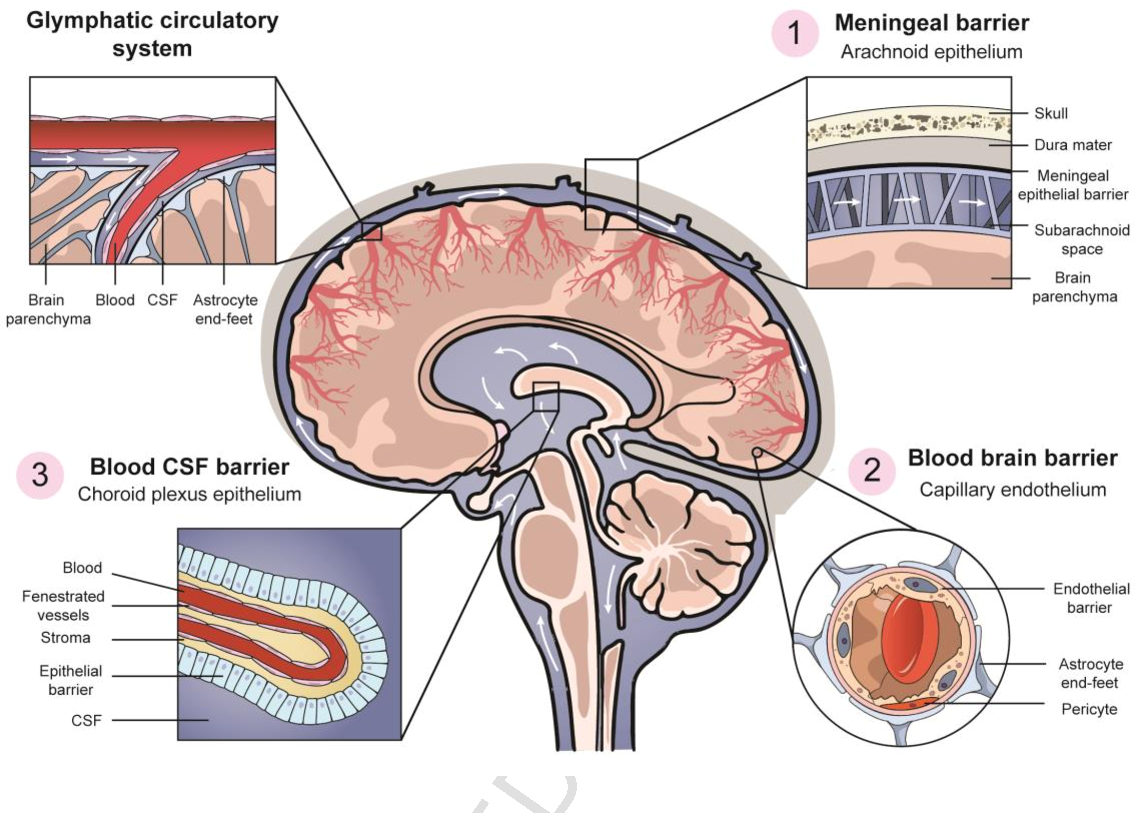 
The literature search strategy employed is illustrated in Fig. This review focuses on current knowledge regarding the relationship between systemic inflammation and the vascular BBB, with three aims: (1) to describe the molecular and cellular components of the BBB that may respond to inflammation, (2) to review how the BBB responds to systemic inflammation, and (3) to define those factors that are known to moderate this relationship. This permissive quality of the vascular BBB may sometimes be taken too far during sustained or overwhelming systemic inflammation or in cases where there is already BBB damage due to neurological disease. This is a set of coordinated physiological and behavioral changes orchestrated by the brain that protects the individual from predators while they are ill (such as via lethargy), enables them to fight the infection (such as via fever and anorexia), and protects the species as a whole (such as via anhedonia and social withdrawal). Second, during systemic inflammation, a number of brain responses occur that have survival value, collectively referred to as sickness behavior. For instance, the brain’s capacity for gluconeogenesis is limited, and glucose supply is heavily reliant on its transport across the vascular BBB via the insulin-independent glucose transporter GLUT-1. First, due to its high level of specialization, the brain depends on the rest of the body for the supply of nutrients and clearance of toxic byproducts of metabolism. There are two main reasons why the barrier cannot be absolute. Although referred to as a “barrier”, this is a misnomer since cells and substances can be exchanged bidirectionally. The vascular blood–brain barrier (BBB) represents the brain’s main interface with its external environment, at which these processes are mostly controlled. On the other hand, a low level of T-cell immunosurveillance is allowed to keep latent viruses under check with minimal inflammatory consequences. Leukocyte entry is highly regulated, with complete suppression of innate immune cell infiltration to avoid acute inflammatory responses in situ, since if neuronal damage occurs, regeneration is slow and limited. The interstitial protein concentration is kept low to minimize cellular proliferation and protein binding of charged ions and neurotransmitters, and to optimize the rheology of extracellular fluids at low pressures in the intracranial compartment. Ionic homeostasis of brain interstitial fluid is essential to enable the maintenance and controlled modulation of transmembrane gradients and movements these are necessary to generate accurate changes in electrical potential locally in synapses, along axons, or in distributed networks. The brain is a delicate organ because neurons require a highly specialized environment to function properly. Further progress is required to define and measure mediators and moderators of the blood–brain barrier’s response to systemic inflammation in order to explain the heterogeneity observed in animal and human studies. These include sex, genetic background, age, pre-existing brain pathology, systemic comorbidity, and gut dysbiosis. Distinct from the molecular and cellular mediators of the blood–brain barrier’s response to inflammation, several moderators influence the direction and magnitude at genetic, system, cellular and molecular levels. 
Here, the molecular and cellular anatomy of the vascular blood–brain barrier is reviewed, first in a healthy context and second in a systemic inflammatory context. Perturbation of neuronal function results in the clinical features of encephalopathy. With increasing degrees and duration of systemic inflammation, the vascular blood–brain barrier becomes more permeable to solutes, undergoes an increase in lymphocyte trafficking, and is infiltrated by innate immune cells endothelial cell damage may occasionally occur. Its primary function is to protect central neurons while signaling the presence of systemic inflammation and infection to the brain to enable a protective sickness behavior response. The vascular blood–brain barrier is a highly regulated interface between the blood and brain.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
